
The concept of synthetic lethality, achieved through PARP inhibitors (PARPi), has ushered in the era of precision therapy for prostate cancer. Clinical studies evaluating PARPi either as monotherapy or in combination with next-generation androgen receptor pathway inhibitors (ARPI), such as PROfound and PROpel, have already produced landmark advances in the treatment of metastatic castration-resistant prostate cancer (mCRPC).The BRCAAway study further evaluated different first-line treatment strategies for patients with mCRPC harboring BRCA1/2 or ATM mutations, including PARPi combined with ARPI, PARPi monotherapy, and ARPI monotherapy. At the 2026 ASCO Genitourinary Cancers Symposium (ASCO-GU), updated results showed that the combination therapy achieved a median overall survival (OS) of 68 months.
Oncology Frontier – Urology Stream invited Prof. Darren Poon from Hong Kong Sanatorium & Hospital to interpret the BRCAAway study.
Q1: In recent years, PARP inhibitors have accumulated a wealth of evidence-based medical data in the field of mCRPC, including the BRCAAway study. First, could you review the design characteristics of this study and the main findings previously reported?
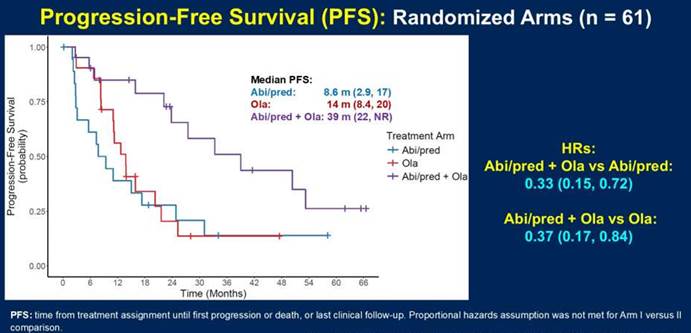
Prof. Darren Poon: The BRACAWAY study is a randomized Phase II trial, which is a biomarker-driven study that evaluate patients with mCRPC disease without prior ARPI or PARP inhibitors. In the germline or somatic testing, they were found to have the BRCA1 or 2 or ATM mutations. So these patients has been enrolled and randomized into one-to-one-to-one fashion into three arms. The first two arms are the monotherapy with either abiraterone prednisone, and the other monotherapy arm is olaparib. And the third arm is olaparib plus abiraterone. So basically, there are three arms and try to evaluate whether the combination approach is the best versus the monotherapy approaches. And one of the very key characteristics of this study is in the monotherapy arms. While the patients have progressed, they could allow for crossover to the other treatments that they haven’t received. Like the patient, if they progress upon abiraterone, they could crossover to olaparib and vice-versa. So previously, the results of this study in regard to the progression-free survival has been released, showing that the combination arm has a very good PFS compared to the monotherapy approaches.
Q2: Following the positive PFS results, the OS outcomes of the BRCAAway study were reported at the ASCO-GU conference. Could you interpret the OS results of this study and their clinical significance?
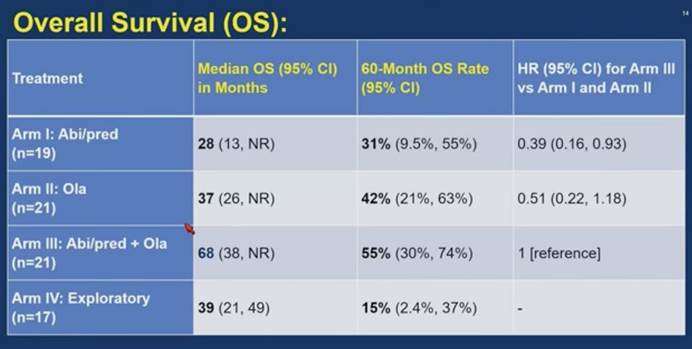
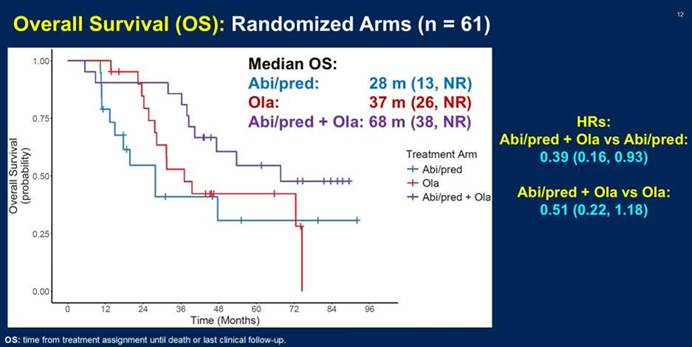
Prof. Darren Poon: Yeah, in ASCO GU 2026, the overall survival data for the BRACAWAY trial has been released and presented. So we can see that the combination arm, that are olaparib plus abiraterone, has the longest median overall survival of up to 68 months, whereas for the monotherapy, such as abiraterone, only 28 months. And for olaparib, the median overall survival is around 38 months. So although this is a randomized Phase II trial and it doesn’t mean to assess the overall survival data as the primary endpoint, but in this study, it clearly showed that the combination arm has a very favorable median overall survival versus the monotherapy approaches.

Q3: The BRCAAway study allowed crossover between the two monotherapy arms, effectively representing a sequential monotherapy treatment model. Based on the crossover data, how do you view the treatment approaches involving PAPRi and NHT?
Prof. Darren Poon: I think the crossover design is a very pragmatic and practical approach and reflect the real-life settings in our clinical practice. Because of the consideration of the toxicities, comorbidities, or the access of the drugs, sometimes the physician will choose to use the monotherapy, either abiraterone or olaparib or PARP inhibitors, and then followed by the sequential use of the other therapies. And I think this is a very practical approach, and in this study, in the BRACAWAY trial, it did show that the patients who crossover to the other drugs, like abiraterone to olaparib, they still have a PSA response and the PFS is also observed with the subsequent therapies, despite the ultimate total PFS or overall survival is not… is still inferior to the combination arm. So therefore, I think this study informed us the different approaches may have a different goals of treatment. For the combination arm, I think the very favorable ultimate overall survival, a deep PSA response, a longer PFS is very favorable, and certainly there will be considerations of additive toxicities. But whereas for the monotherapies, I think for patients who are concerned of the toxicities or the other considerations like comorbidities, I think sequential approaches with different monotherapies is also a very reasonable approach.
Q4: Taking a comprehensive look at the PROfound, PROpel, and BRCAAway studies, how do you assess the clinical application value of APRPi in the mCRPC field? Additionally, what impact does it have on precision treatment for mCRPC?
Prof. Darren Poon: I think these three important landmark trials—the PROpel, the PROfound, and the BRACAWAY trial—inform us that the genomic testing is important in the treatment paradigm for the metastatic prostate cancer in different settings. For example, in the PROfound trial, it established the role of PARP inhibitors in mCRPC patients who had prior ARPI and chemotherapy. And in the PROpel trial, it established the role of combination treatments with the olaparib together with abiraterone had a significant improvement in PFS and OS versus the abiraterone alone. Whereas in the BRACAWAY trial, we understand that the combination approaches with the PARP inhibitors together with abiraterone has a more favorable overall survival and PFS compared to the sequential use of the monotherapies. So all these trials showing that the genomic testing is important, and if we could find out patients who harbor the BRCA1, 2 mutations or even the other HRR mutations, these patients will be benefit from the additions of PARP inhibitors, either monotherapy or in combinations with the ARPI in different settings of the mCRPC disease. So therefore, we should be bear in mind genomic testing is nowadays a very important treatment approach for the management of mCRPC patients.



