
Although CD19 CAR-T therapy achieves high remission rates in relapsed/refractory B-cell acute lymphoblastic leukemia (R/R B-ALL), relapse driven by CD19 antigen loss poses a major challenge for MRD monitoring and long-term disease management. Identifying more stable biomarkers has therefore become essential for improving response assessment and prognostic evaluation. At the 52nd EBMT Annual Meeting (March 22–25, 2026, Madrid, Spain), the team led by Prof. Hui Wang and Prof. Man Chen from Lu Daopei Hospital presented their work on CD72-gated MRD analysis. Their findings demonstrate the value of CD72 in overcoming the limitations of CD19-based monitoring and in predicting post-transplant outcomes.
This article features a detailed overview provided by Prof. Man Chen.
Study Background
CD19 CAR-T cell therapy has achieved remarkable success in B-cell malignancies. However, some patients experience relapse due to reduced or complete loss of CD19 antigen expression, leading to CD19-negative relapse.
This phenomenon poses significant challenges for post-treatment monitoring and highlights the urgent need for alternative biomarkers.
CD72, a member of the C-type lectin superfamily, is broadly expressed across B-cell developmental stages and represents a promising candidate both as a therapeutic target and as a monitoring marker.
This study aimed to evaluate the application of multiparameter flow cytometry–based MRD (MFC-MRD) across the entire CAR-T-to-transplant treatment course, assess the feasibility of CD72 as a B-lineage target, and identify key factors influencing outcomes in R/R B-ALL.
Methods
Correlation analyses were performed in 66 B-ALL patients (25 MRD-negative and 41 MRD-positive), comparing CD72- and CD19-based gating strategies.
In addition, CD72 expression was retrospectively analyzed in 129 patients (71 B-ALL, 41 AML, 17 T-ALL) to evaluate its specificity for B cells.
Key parameters included CD72 expression rate, median fluorescence intensity (MdFI), and the MdFI ratio relative to control T lymphocytes (MdFIR).
To further assess specificity, CD72 expression was also evaluated in 32 patients with B-cell non-Hodgkin lymphoma (B-NHL) and 10 patients with multiple myeloma (MM).
For clinical validation, 129 patients with R/R B-ALL treated between January 2021 and December 2022 at Hebei Yanda and Beijing Lu Daopei Hospitals were retrospectively analyzed. All patients were enrolled according to clinical trial criteria (ChiCTR-IIh-16008711; NCT03173417).
Patients were divided into two cohorts based on pre–CAR-T MRD levels measured by CD72-gated MFC-MRD:
- Cohort 1: MRD ≤1%
- Cohort 2: MRD >1%
All patients underwent hematopoietic stem cell transplantation (HSCT) and were followed until January 2025.
MRD monitoring time points included:
- Pre–CAR-T (day 0)
- Day 15 and day 28 post–CAR-T
- Every 30 days until transplantation
- Monthly during the first 3 months post-transplant
- Every 3 months from months 3–12
- Every 6 months thereafter
Samples included bone marrow, cerebrospinal fluid, and extramedullary tissues. CD19-positive B-cell recovery was also recorded.
Results
1. Concordance Between CD72 and Conventional Gating
CD72 gating showed excellent correlation with CD19 and cCD79a-based methods.
In 25 patients in complete remission, results were consistent across methods, with strong correlation in normal B-cell proportions:
- CD72/CD19: R² = 0.9936, y = 0.9363x + 0.0418
- CD72/cCD79a: R² = 0.9907, y = 0.9297x + 0.071
In 41 non-remission patients, malignant B-cell proportions were also highly correlated:
- CD72/CD19: R² = 0.9993, y = 0.9308x + 0.1675
- CD72/cCD79a: R² = 0.9991, y = 0.9833x + 0.1393
2. Specificity of CD72 Expression
CD72 positivity rates:
- AML: 29.27% (3/41 positive, 4 weakly positive, 5 partially positive, 29 negative), MdFIR 1.38 (2.77, 6.35)
- T-ALL: 23.53% (1 weakly positive, 3 partially positive, 13 negative), MdFIR 1.11 (0.99, 3.27)
- B-ALL: 95.77% positive, MdFIR 41.43 (20.65, 79.60)
In B-ALL samples, normal B cells showed a significantly higher CD72 MdFIR of 88.24 (49.61, 163.37), with a significant difference compared to leukemic cells (P < 0.001).
CD72 expression in B-ALL was significantly higher than in AML and T-ALL (P < 0.001).
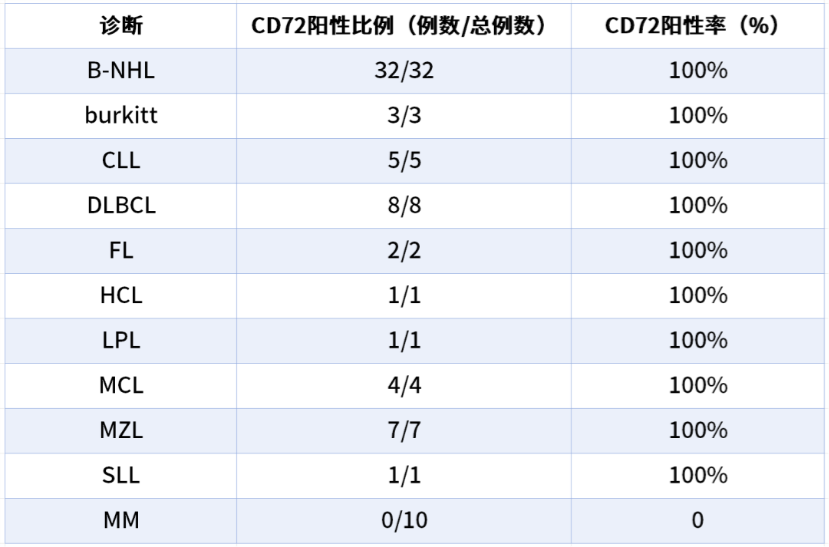
3. CD72 Expression in B-NHL and MM
CD72 expression was analyzed in 32 B-NHL and 10 MM patients (Table 1), confirming its relevance across B-cell malignancies.
4. Clinical Follow-up and Application
Among 129 patients:
- Cohort 1 (MRD ≤1%): 51 patients, median MRD 0.05% (0.003%–0.93%)
- Cohort 2 (MRD >1%): 78 patients, median MRD 5.3% (1.1%–77.4%)
Baseline characteristics were balanced between groups.
With a median follow-up of 38.5 months (1.2–48.3):
- 30 deaths occurred, 70% within the first year
- 100-day transplant-related mortality: 3.9%
- 3-year OS: 76.6%
- 3-year cumulative MRD relapse incidence: 12.46% (25% CD19-negative relapse)
B-cell recovery:
- No difference in recovery rate or proportion between groups
- Cohort 1 showed significantly earlier recovery of both CD19-positive and CD19-negative B cells
Prognostic analysis:
- 3-year OS: 88.1% (Cohort 1) vs. 69.2% (Cohort 2)
- 3-year relapse incidence: 3.96% vs. 17.95%
- NRM: trend lower in Cohort 1 (5.92% vs. 17.95%)
Multivariate analysis identified:
- KMT2A rearrangement
- IKZF1 mutation
- TP53 mutation
- Pre–CAR-T MRD ≥1%
as independent adverse prognostic factors for OS, LFS, or NRM.
Timing of transplantation (≤45 days, 45–60 days vs. >60 days) and CAR-T dose had no significant impact on outcomes.
Conclusion
A novel MFC-MRD system based on cytoplasmic CD72 gating provides a reliable method for MRD monitoring in B-ALL.
This approach demonstrates strong clinical utility in prognostic assessment, early relapse detection, and treatment decision-making. Further optimization—such as increasing monitoring frequency, extending follow-up duration, and incorporating additional sample types (e.g., CSF and extramedullary lesions)—may enhance its role in personalized disease management.
Investigator Profiles

Hui Wang, PhD Lu Daopei Hospital
Prof. Wang is Deputy Director of the Beijing Lu Daopei Institute of Hematology and Vice Director (vice president level) of the Clinical Laboratory at Lu Daopei Hospital. She has over 25 years of experience in flow cytometry, has issued over one million clinical reports, holds multiple Chinese and U.S. patents, and has authored more than 60 SCI and core journal publications.

Man Chen, MD Lu Daopei Hospital
Prof. Chen is Head of the Flow Cytometry Laboratory in the Department of Pathology and Laboratory Medicine at Beijing Lu Daopei Hospital. She has over 14 years of experience in flow cytometry diagnostics and leads CAP-accredited laboratory operations. She has published 23 SCI/core papers, holds multiple patents, and has contributed to national guidelines and consensus statements.


