Renal cell carcinoma (RCC) is one of the most common malignant tumors of the urinary system, among which approximately 75% are clear cell renal cell carcinoma (ccRCC). Loss of the VHL gene is the key molecular event driving this disease. VHL inactivation places tumor cells in a persistent state of “pseudohypoxia,” continuously activating HIF-2α-centered oncogenic transcriptional programs that promote angiogenesis, cell proliferation, and metabolic reprogramming.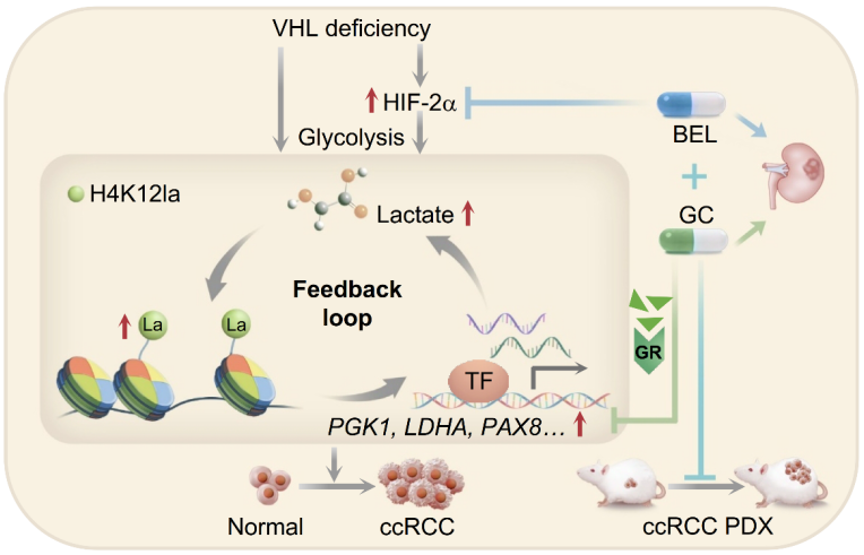
HIF-2α inhibitors, such as belzutifan, have already entered clinical practice. However, their objective response rate remains limited at around 25%. Identifying agents that can synergize with HIF-2α inhibitors to enhance efficacy has therefore become an important research focus.
On April 1, the urology team led by Professor Kan Gong at Peking University First Hospital, together with the team of Sun Luyang from Peking University Health Science Center and the team of Qing Zhang from UT Southwestern Medical Center, published an important study in the international top-tier journal Signal Transduction and Targeted Therapy (impact factor 52.7).
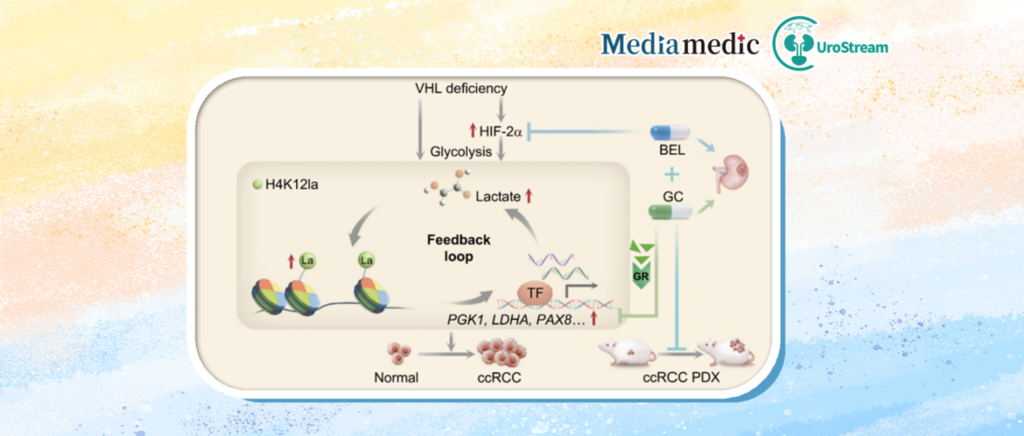
The paper, titled “Glucocorticoids elevate clear cell renal cell carcinoma sensitivity to HIF-2α inhibitors by suppressing H4K12 lactylation,” systematically described the mechanistic role of H4K12la in VHL-deficient renal cancer and uncovered the “H4K12la–PGK1–lactate” positive feedback loop.
The study found that glucocorticoids, particularly dexamethasone, can suppress this loop and produce significant synergistic antitumor effects when combined with the HIF-2α inhibitor belzutifan. These findings provide new mechanistic and therapeutic evidence supporting combination treatment strategies.
The research may offer a new precision treatment direction for VHL-deficient renal cancer and has the potential to optimize current targeted therapies while improving long-term patient benefit.

Discovery of a Vicious Cycle: From “Waste” Lactate to “Fuel” for Epigenetic Regulation
Approximately 70% of patients with clear cell renal cell carcinoma harbor VHL gene loss. VHL deficiency reprograms cellular metabolism from mitochondrial oxidative phosphorylation toward highly glycolysis-dependent metabolism, accompanied by substantial lactate accumulation.
Lactate was once considered merely a metabolic waste product. However, increasing evidence suggests that lactate can directly “write” metabolic status into chromatin through epigenetic mechanisms such as histone lactylation, thereby continuously reinforcing pro-tumor transcriptional programs. These findings imply the existence of a lactate-driven metabolic regulatory axis in renal cancer.
Therefore, clarifying how lactate promotes ccRCC progression and identifying drugs capable of disrupting this lactate-driven pathway have become important research priorities.
Through immunohistochemical analysis, the investigators first found that H4K12la levels were markedly elevated in clear cell renal cell carcinoma and closely associated with higher tumor grade and poorer patient prognosis.
Using integrated multi-omics analyses including ATAC-seq and CUT&Tag, the researchers further demonstrated that in VHL-deficient tumor cells:
Global chromatin accessibility increased
H4K12la became significantly enriched at promoter regions
Oncogenic genes including the glycolytic regulator PGK1 and renal lineage transcription factor PAX8 were transcriptionally activated
The glycolytic enzyme PGK1 further increased lactate production, which in turn promoted additional H4K12la accumulation.
These findings revealed that H4K12la may not only serve as a potential prognostic biomarker for renal cancer, but may also drive ccRCC progression through establishment of the “H4K12la–PGK1–lactate” positive feedback loop.
Old Drug, New Use: Glucocorticoids Become “Cycle Breakers”
The research team subsequently performed high-content screening of 2,468 FDA-approved compounds and discovered that multiple glucocorticoids (GCs) significantly reduced H4K12la levels in VHL-deficient renal cancer cells, with dexamethasone (DEX) showing the strongest effect.
Mechanistically, DEX activated the glucocorticoid receptor (GR), promoting GR nuclear translocation and binding to promoter regions of multiple glycolysis-related genes. This transcriptionally suppressed glycolytic target genes and reduced lactate production.
In animal models, combined treatment with DEX and the HIF-2α inhibitor belzutifan inhibited tumor growth significantly more effectively than either agent alone.
Summary
This study demonstrates that glucocorticoids can precisely disrupt the lactate-driven “H4K12la–PGK1–lactate” epigenetic positive feedback loop through the GR signaling axis, while simultaneously complementing HIF-2α inhibitors at the hypoxia signaling level.
Together, these effects establish a novel “dual-brake” synergistic treatment model combining metabolic-epigenetic regulation with suppression of oncogenic transcriptional programs.
This strategy may help overcome current limitations of single-agent therapy, including limited treatment depth and resistance development, and could provide a clinically translatable new pathway for precision treatment of VHL-deficient renal cancer. It may also offer broader insight into combination strategies for metabolically driven tumors.

Professor Kan Gong from Peking University First Hospital, Sun Luyang from Peking University Basic Medical Sciences, and Qing Zhang from UT Southwestern Medical Center served as corresponding authors.

Dr. Kenan Zhang from Peking University First Hospital was the first author, while Associate Professor Lin He from Peking University served as co-first author and corresponding author.


