
Editor’s Note: Platinum-resistant recurrent ovarian cancer (PROC) remains a major clinical challenge, characterized by poor overall prognosis, limited effective treatment options, high unmet medical need, and suboptimal safety profiles. Innovative treatment strategies are urgently needed.On April 19, 2026 (local time), at the Annual Meeting of the American Association for Cancer Research (AACR), Professor Tao Zhu from Zhejiang Cancer Hospital presented results from a first-in-human Phase I study of the CLDN6-targeting antibody–drug conjugate (ADC) QLS5132 in patients with PROC. The study demonstrated an objective response rate (ORR) exceeding 50% alongside a favorable safety profile. Notably, preliminary findings suggest that CLDN6 expression levels may not significantly impact treatment efficacy, indicating potential applicability across a broad patient population.
Oncology Frontier conducted an exclusive interview with Professor Tao Zhu to discuss the current challenges in PROC management, the unique characteristics of the CLDN6 target, and the clinical implications of the QLS5132 study.

01 | Clinical Challenges in Platinum-Resistant Ovarian Cancer
Oncology Frontier: Platinum resistance and recurrence remain major issues following first-line therapy in advanced ovarian cancer. Based on your clinical experience, what are the key challenges faced by these patients?
Professor Tao Zhu:
The development of PROC often signals limited treatment options, suboptimal therapeutic efficacy, and poor prognosis, making it one of the most pressing challenges in gynecologic oncology.
First, patient outcomes are extremely unfavorable. Ovarian cancer is characterized by high rates of recurrence and progression. With each relapse, tumor sensitivity to chemotherapy progressively declines, eventually leading to platinum resistance. Once this stage is reached, the efficacy of conventional chemotherapy diminishes sharply. With non-platinum single-agent chemotherapy, the objective response rate is typically only 10%–20%, and meaningful tumor shrinkage is rarely achieved. Median progression-free survival (PFS) is approximately 3–4 months, reflecting rapid disease progression, while median overall survival (OS) is around 13 months, underscoring the urgent need for improvement.
Even when combined with anti-angiogenic agents such as bevacizumab, clinical benefit remains limited, and current approaches struggle to break through this therapeutic ceiling.
Second, available treatment options remain scarce. Beyond conventional chemotherapy, only a small number of ADCs have been approved for PROC, including those targeting folate receptor alpha (FRα) and HER2. However, these therapies depend heavily on target expression and are therefore limited to biomarker-positive patients. In addition, treatment-related toxicities can affect tolerability and quality of life, further constraining clinical use.
Overall, PROC patients face multiple challenges, including poor prognosis, limited effective therapies, high unmet medical need , and the need for improved safety profiles. There is a clear need for novel agents with innovative mechanisms, broader applicability, higher efficacy, and better tolerability to meaningfully improve survival and quality of life.
02 | CLDN6 as a Promising Therapeutic Target
Oncology Frontier: With the development of precision medicine, CLDN6 has gained increasing attention and is now recommended as a testing target in the 2026 NCCN guidelines. Could you elaborate on its characteristics and clinical value?
Professor Tao Zhu:
The primary value of CLDN6 lies in its tumor-specific expression pattern, which distinguishes it from most solid tumor targets. In normal adult tissues, CLDN6 is essentially silenced and is not detectable in key organs such as the ovary, lung, stomach, and liver. It is only transiently expressed during early embryonic development, resulting in minimal risk of off-target toxicity.
In contrast, CLDN6 is highly expressed in a range of malignancies. In advanced, platinum-resistant high-grade serous ovarian cancer—where the clinical need is greatest—the positivity rate reaches 60%–70%, significantly higher than in treatment-naive cases. Moreover, its expression remains relatively stable in recurrent and metastatic lesions.
This ideal distribution—high expression in tumors and near absence in normal tissues—provides a strong foundation for targeted therapy. It allows for precise tumor targeting while minimizing damage to healthy tissues, thereby achieving both strong antitumor activity and favorable safety.
Building on these properties, CLDN6 has become a key focus for multiple innovative therapeutic platforms, including ADCs, bispecific T-cell engagers (BiTE), and CAR-T/CAR-NK therapies. These approaches aim to address the unmet needs in PROC, where conventional chemotherapy is ineffective and immunotherapy response rates remain low.
Future research will focus on clarifying the relationship between CLDN6 expression and treatment response, optimizing combination strategies, and exploring resistance mechanisms related to tumor heterogeneity and target loss, with the goal of translating this promising biology into meaningful clinical benefit.
03 | Study Design of the First-in-Human QLS5132 Trial
Oncology Frontier: At this AACR meeting, you presented the first-in-human study of QLS5132. Could you describe the study design and how safety and early efficacy were balanced?
Professor Tao Zhu:
The study design integrates accelerated titration and Bayesian optimal interval (BOIN) methodologies to balance efficiency, safety, and flexibility. It follows a strategy of first focusing on high-priority populations and then expanding to other tumor types, with dose escalation and expansion phases closely integrated.

△First-in-Human Study of QLS5132
During dose escalation, the BOIN design, combined with a backfilling strategy, enabled efficient identification of the maximum tolerated dose (MTD) or recommended phase II dose (RP2D), while simultaneously capturing early signals of antitumor activity. This approach ensures robust safety assessment while maintaining development efficiency.
Patient enrollment initially focused on tumor types with high CLDN6 expression and urgent clinical need, particularly PROC, to facilitate early proof of concept. Notably, CLDN6 expression was not required for enrollment at this stage, and the primary endpoint was safety, including dose-limiting toxicities (DLTs), adverse events, and tolerability.
After determination of the RP2D, the study enters the expansion phase, focusing on tumor types with demonstrated activity, particularly PROC, while also exploring efficacy in other malignancies such as lung, gastric, and endometrial cancers.
This design—broad early exploration followed by focused expansion—ensures a scientifically rigorous and efficient clinical development pathway.
04 | Clinical Implications and Future Directions
Oncology Frontier: Based on the preliminary results, what are the key clinical implications, and what future directions should be pursued?
Professor Tao Zhu:
QLS5132, as a next-generation CLDN6-targeting ADC, has demonstrated highly promising early clinical activity in PROC, offering new hope in a setting with significant unmet need.
First, the study demonstrates a meaningful efficacy improvement, with an ORR exceeding 50% and a duration of response exceeding six months in the early cohort, surpassing existing non-platinum chemotherapy options.
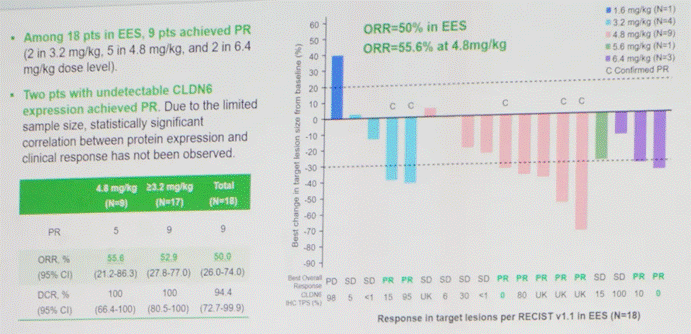
△Best Overall Response to QLS5132
Second, the safety profile is favorable. Most adverse events were grade 1–2, with no interstitial lung disease or ocular toxicity observed, and low rates of rash and peripheral neuropathy. This suggests a good balance between efficacy and tolerability.
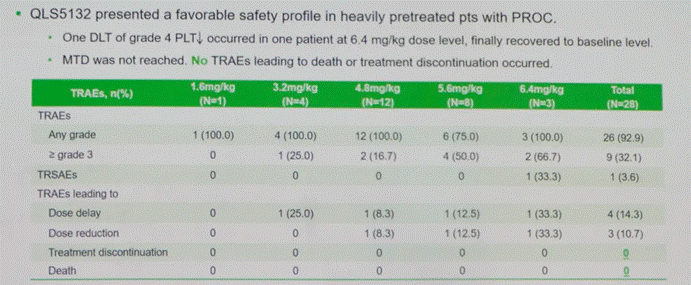
△Safety Profile of QLS5132
Finally, preliminary data suggest that efficacy may not depend strongly on CLDN6 expression levels, raising the possibility of broader patient applicability without strict biomarker selection, pending confirmation in larger studies.
Future development will focus on conducting pivotal Phase III trials in PROC and exploring earlier-line treatment settings, including platinum-sensitive and first-line populations, to expand the clinical benefit.



Expert Profile

Tao Zhu
Zhejiang Cancer Hospital
Vice President, Zhejiang Cancer Hospital
Deputy Director, Zhejiang Cancer Diagnosis and Treatment Quality Control Center
Vice Chairman, Zhejiang Cancer Foundation
Vice Chair, Chinese Anti-Cancer Association Integrative Gynecologic Oncology Committee
Chair, Cervical Cancer Integrated Prevention and Screening Committee, Chinese Anti-Cancer Association
Vice Chair, The Society of Cervical Cancer, Chinese Anti-Cancer Association
Chair, Gynecologic Oncology Committee, Zhejiang Medical Doctors Association
Visiting Scholar, MD Anderson Cancer Center
Principal Investigator (PI), sub-centers of the LACC and SENTICOL III studies
