
The 2026 European Association of Urology Annual Congress (EAU26), held in London, once again served as a premier platform showcasing cutting-edge advances in urologic oncology. At this year’s meeting, Prof. Kan Gong and his team from Peking University First Hospital presented a series of impactful studies focusing on von Hippel–Lindau (VHL) syndrome–associated renal cell carcinoma in the Chinese population. In this exclusive discussion, Prof. Gong provides an in-depth interpretation of their findings, highlighting population-specific mutation patterns, genotype–phenotype correlations, and emerging insights with direct clinical relevance.
Distinct Mutation Spectrum in Chinese Patients
The study represents one of the largest retrospective cohorts of VHL syndrome to date, including 1,172 Chinese patients. Analysis of this large dataset revealed a mutation spectrum with clear population-specific characteristics.
Missense mutations were the most prevalent, accounting for approximately 50% of cases, followed by large fragment deletions at nearly 18%. These findings suggest that functional alteration of the VHL protein—rather than complete loss—is the dominant pathogenic mechanism in this population.
A particularly notable discovery was the identification of codon 167 as a high-frequency mutation hotspot among Chinese patients. Carriers of this mutation demonstrated a significantly increased risk of pheochromocytoma, while showing a relatively lower risk of mortality associated with hemangioblastomas. This genotype-specific clinical pattern provides valuable guidance for individualized surveillance and management strategies.
Overall, the findings indicate that pathogenic mutations are concentrated in key functional domains of the VHL protein, particularly within the Elongin C binding region, reinforcing the biological relevance of these structural sites.
Clear Genotype–Phenotype Correlations
The study further confirms that mutation type strongly correlates with clinical presentation in VHL syndrome, offering a reliable framework for risk stratification.
Truncating mutations, which typically result in complete loss of VHL protein function, are characteristic of type 1 VHL syndrome. These patients are more prone to developing retinal and central nervous system hemangioblastomas as well as clear cell renal cell carcinoma, while having a lower risk of pheochromocytoma.
In contrast, missense mutations tend to preserve partial protein function and are associated with type 2 VHL syndrome. These patients carry a significantly higher risk of pheochromocytoma and can be further subdivided into three clinical subtypes. Type 2A is characterized by pheochromocytoma and hemangioblastoma with relatively low renal cancer risk. Type 2B involves high risks across pheochromocytoma, hemangioblastoma, and renal cancer. Type 2C presents predominantly as isolated pheochromocytoma.
From a clinical course perspective, patients with truncating mutations typically develop hemangioblastomas early, followed by renal manifestations in early adulthood. In contrast, those with missense mutations are more likely to present with pheochromocytoma at an earlier stage.
Mechanistically, these differences are thought to arise from varying degrees of disruption in hypoxia-inducible factor (HIF) regulation, particularly involving HIF-1α and HIF-2α signaling pathways.
Refining Risk Stratification Beyond Traditional Classification
Beyond conventional classification systems, the team identified additional layers of genotype–phenotype association by stratifying missense mutations according to their functional domains.
Missense mutations located within the HIF-α binding region were associated with a lower risk of pheochromocytoma but a higher likelihood of hemangioblastoma. This nuanced classification provides a more refined approach to clinical risk prediction and highlights the importance of functional domain–based mutation analysis.
Focusing on missense mutations offers particular value in the Chinese population, given their high prevalence and the relative precision with which single amino acid substitutions can be linked to specific phenotypic outcomes. These findings contribute to the optimization of individualized risk assessment models and genetic counseling strategies.
Metabolomics Insights into Early Detection and Anticipation
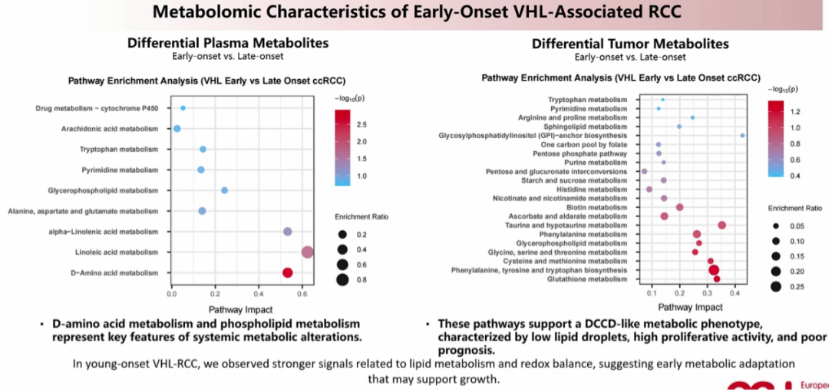
A particularly innovative aspect of the research lies in its exploration of metabolomic differences between early-onset and late-onset patients, as well as between successive generations within affected families.
The study identified consistent and reproducible metabolic alterations in early-onset patients and younger-generation individuals, detectable in plasma and tumor tissue samples. These metabolic signatures appear to precede radiologically detectable lesions, suggesting their potential as early biomarkers for disease detection.
Clinically, this opens the possibility of developing blood-based metabolomic tools for risk stratification, enabling earlier identification of high-risk individuals and more precise timing of surveillance and intervention.
Furthermore, these findings offer new insights into the phenomenon of genetic anticipation observed in VHL syndrome. Altered steroid hormone metabolism, particularly involving the androgen–androgen receptor (AR) axis, was identified as a potential contributing factor. Experimental validation demonstrated that AR signaling promotes tumor cell proliferation and drives lipid metabolic reprogramming.
This suggests that earlier disease onset in subsequent generations may not be solely attributable to genetic inheritance but may also involve a predisposing metabolic and endocrine microenvironment.
Looking Ahead
Prof. Gong emphasizes that while these findings provide important biological and clinical insights, further validation through large-scale prospective studies will be essential before translation into routine clinical practice.
Nonetheless, this work represents a significant step forward in understanding VHL syndrome within the Chinese population and underscores the importance of integrating genomics, metabolomics, and clinical data to advance precision oncology.

Prof. Kan Gong


