
In real-world clinical practice, a considerable proportion of patients with multiple myeloma (MM) are unable to tolerate autologous stem cell transplantation (ASCT) due to advanced age, comorbidities, or poor performance status, creating an urgent need for more effective therapeutic strategies. On February 27, 2026, a research paper entitled Phase II Study of BCMA Chimeric Antigen Receptor T-Cell Therapy in Patients With Newly Diagnosed Multiple Myeloma Ineligible for or Not Proceeding to Autologous Stem-Cell Transplantation (CAREMM-001) by a Chinese research team was published online in the Journal of Clinical Oncology (IF=43.4). This study provides the first prospective clinical evidence for the field, confirming that the upfront application of B-cell maturation antigen (BCMA) chimeric antigen receptor T-cell (CAR-T) therapy as first-line treatment for patients with newly diagnosed multiple myeloma (NDMM) who are ineligible for or not proceeding to transplantation—including those with high-risk disease—achieves an impressive 100% minimal residual disease (MRD) negativity rate and a 94.4% complete response rate (CRR) with a favorable safety profile. This novel therapeutic approach is expected to establish a new paradigm for MM treatment. This article summarizes the key findings of this landmark study for clinical reference.
Multiple myeloma is a common malignant plasma cell disorder of the hematologic malignancies, and ASCT serves as the cornerstone of first-line treatment for eligible patients with newly diagnosed disease, significantly improving long-term disease control outcomes. However, in real-world clinical settings, a substantial proportion of NDMM patients cannot undergo or tolerate ASCT due to advanced age, poor performance status, underlying comorbidities, or failed stem cell mobilization, highlighting an urgent clinical need for safer and more effective first-line therapeutic strategies.
Previous studies have demonstrated that BCMA-targeted CAR-T therapy exhibits remarkable efficacy and a manageable safety profile in patients with relapsed/refractory multiple myeloma (RRMM). Its clinical feasibility has also been verified in elderly or frail MM patients, laying a solid foundation for the upfront use of this therapy in first-line treatment.
On February 27, 2026, the CAREMM-001 study by a Chinese research team was published online in the Journal of Clinical Oncology. This study marks the first application of BCMA CAR-T therapy as first-line treatment for transplant-ineligible NDMM patients, which not only significantly deepens treatment responses but also avoids the severe toxicities associated with multiple cycles of chemotherapy. It provides pivotal prospective clinical evidence for first-line treatment in this patient population and confirms that upfront BCMA CAR-T therapy achieves 100% MRD negativity and 94.4% CRR in transplant-ineligible NDMM patients (including those with high-risk features) with a favorable safety profile, which is expected to reshape the clinical treatment paradigm of multiple myeloma. The following sections summarize the core findings of this study to provide insights for clinical practice.
Study Design: Validating the Feasibility of Frontline BCMA CAR-T Therapy
CAREMM-001 is an open-label, single-arm, phase II clinical trial (NCT05860036) conducted at the Institute of Hematology and Blood Diseases Hospital (Chinese Academy of Medical Sciences and Peking Union Medical College, Tianjin, China), with a study period from April 4, 2023, to December 26, 2024. The primary objective of the trial was to evaluate the efficacy and safety of BCMA CAR-T therapy in NDMM patients who are ineligible for or not proceeding to ASCT. A total of 40 eligible patients with newly diagnosed transplant-ineligible MM were ultimately enrolled, among whom 36 completed BCMA CAR-T cell infusion.
The primary endpoint was the MRD negativity rate (at a sensitivity threshold of 10⁻⁵) at 3 months post-infusion. Secondary endpoints included CRR, progression-free survival (PFS), overall survival (OS), and duration of response (DOR). Treatment-emergent adverse events (TEAEs) were also monitored to assess safety. All enrolled patients received a sequential treatment strategy consisting of short-course induction therapy, BCMA CAR-T cell infusion, consolidation therapy, and maintenance therapy. The data cutoff date for the study was September 1, 2025, with a median follow-up of 15.8 months (range: 4.3–26.0 months) post-infusion for the infused cohort.
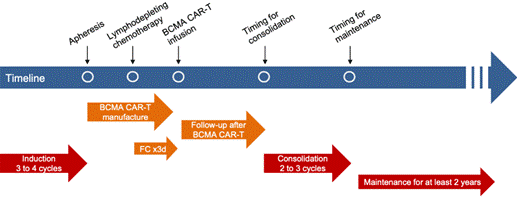
Treatment Regimen: Stepwise Sequential Therapy
The CAREMM-001 study established a standardized stepwise sequential treatment regimen, with individualized adjustments at each stage based on the patient’s clinical status. The detailed regimen is as follows:
- Induction therapy: All patients received 3 to 4 cycles of induction therapy, based on the standard VRd regimen (bortezomib + lenalidomide + dexamethasone) or alternative regimens containing daratumumab (D-VRd, D-Rd), to reduce tumor burden for subsequent CAR-T therapy.
- CAR-T cell manufacturing and infusion: Following induction therapy, leukapheresis was performed for CAR-T cell manufacturing. T lymphocytes were isolated using anti-CD3 immunomagnetic beads and transduced with a lentiviral vector to generate second-generation BCMA-targeted CAR-T cells. Patients without active infection received lymphodepletion chemotherapy (fludarabine 30 mg/m² plus cyclophosphamide 300 mg/m² on days -5, -4, and -3), followed by a single intravenous infusion of BCMA CAR-T cells on day 0 at a target dose of 3.0×10⁶ cells/kg, with a minimum acceptable dose of 1.0×10⁶ cells/kg.
- Consolidation therapy: Initiated at 3 months post-infusion, the regimen was consistent with induction therapy excluding glucocorticoids, with 2 to 3 cycles completed to further deepen disease remission.
- Maintenance therapy: After consolidation therapy, lenalidomide maintenance was administered for a planned minimum of 2 years, until disease progression, unacceptable toxicity, or patient withdrawal.
Patient Characteristics: Confronting High-Risk Clinical Challenges
The study enrolled a patient population with prominent features of advanced age, frailty, and high-risk disease, which closely reflects the real-world patients facing clinical treatment dilemmas and thus renders the study’s efficacy data more compelling.
In the infused cohort (n=36), the median age was 68 years (range: 46–75 years), with 85.0% of patients aged ≥65 years (25% of whom were ≥70 years). Additionally, 19.4% of patients had an Eastern Cooperative Oncology Group (ECOG) performance status score of 2, and 2.8% had a score of 3; 27.8% of patients were classified as frail according to the International Myeloma Working Group Frailty Index (IMWG-FI).
In terms of high-risk disease features, the population exhibited a high prevalence of adverse prognostic factors: 37.1% were ultra-high-risk (UHR), including 11.1% with extramedullary disease, 8.3% with circulating plasma cells ≥2%, and 21.2% with double-hit cytogenetics (≥2 cytogenetic abnormalities including gain(1q), del(17p), t(4;14), etc.). Moreover, 25.7% of patients were classified as Revised International Staging System (R-ISS) stage III, 48.6% as Consensus Genomic Staging (CGS) high-risk, and the incidence of classic high-risk cytogenetic abnormalities reached 39.4%.
Overall, the enrolled patients were all transplant-ineligible NDMM patients with high clinical treatment difficulty, setting rigorous and clinically meaningful conditions for verifying the efficacy of BCMA CAR-T therapy.
Efficacy Outcomes: Deep and Durable Remission
The CAREMM-001 study yielded striking efficacy results: BCMA CAR-T therapy significantly deepened disease remission in transplant-ineligible NDMM patients with durable responses, even achieving deep remission in ultra-high-risk patients and breaking through the efficacy bottleneck of traditional therapies.
For the primary endpoint, all 36 patients who received CAR-T infusion achieved MRD negativity at 3 months post-infusion (95% CI: 90.3 to 100.0), with a median time to MRD negativity of only 28 days (range: 28–33 days). During a median follow-up of 15.8 months post-infusion, no MRD recurrence was observed in any patient: 26 patients maintained MRD negativity for ≥12 months, 31 for ≥6 months, and the remaining 5 patients remained MRD-negative for the entire follow-up period, confirming the excellent durability of the deep molecular remission induced by this therapy.
For secondary endpoints, the disease response rate increased continuously with treatment: the pre-infusion CRR was only 33.3% (95% CI: 18.6 to 51.0), rising to 69.4% (95% CI: 51.9 to 83.7) at 3 months post-infusion and further to 94.4% (95% CI: 81.3 to 99.3) at the last follow-up. The median time to complete response was 94 days (range: 28–416 days) post-infusion. Notably, all patients who received CAR-T infusion achieved at least a very good partial response (VGPR), and 100% of patients with suboptimal responses pre-infusion experienced deepened remission after CAR-T therapy. As of the data cutoff date, no disease progression or death was reported, and the median PFS, OS, and DOR were not yet estimable, with existing data showing a promising trend in survival benefit.
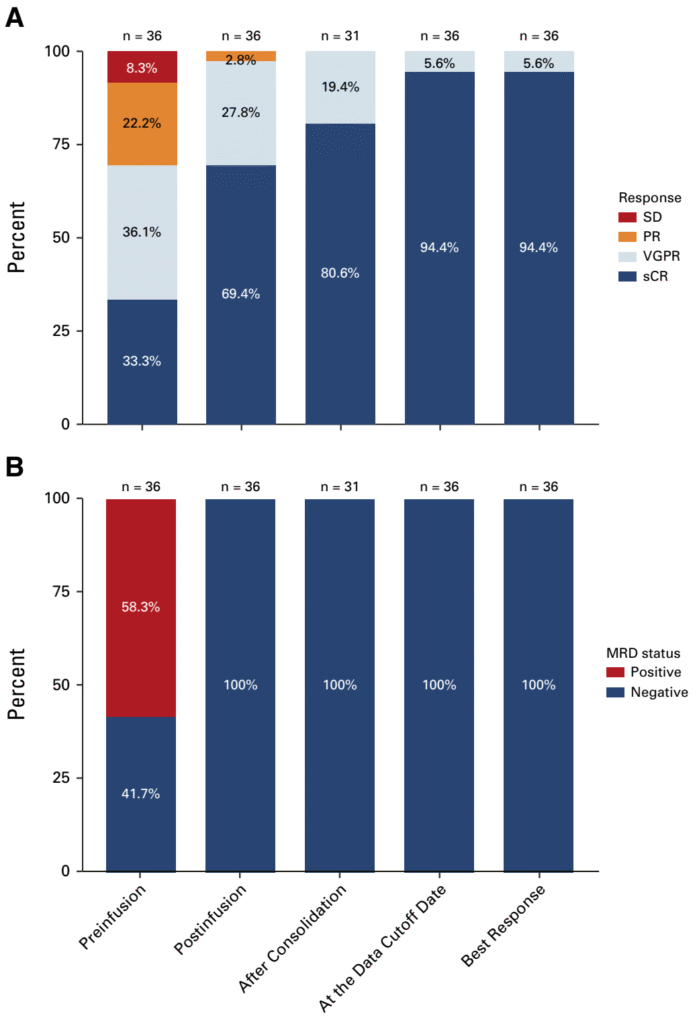
In the intention-to-treat (ITT) population (n=40), the MRD negativity rate at 3 months post-infusion was 90.0% (95% CI: 76.3 to 97.2) and the CRR was 62.5% (95% CI: 45.8 to 77.3), still maintaining a high response level. Subgroup analysis showed that 100% MRD negativity was achieved at 3 months post-infusion regardless of whether patients received a VRd-based or daratumumab-containing induction regimen, indicating that the choice of induction regimen does not affect the core efficacy of BCMA CAR-T therapy. Furthermore, more sensitive next-generation sequencing (NGS) for MRD assessment (sensitivity threshold of 10⁻⁶) revealed that the pre-infusion NGS-MRD negativity rate was only 26.9%, increasing to 92.3% at 3 months post-infusion. Soluble BCMA (sBCMA) levels were reduced by 96.4% from baseline, further confirming the profound anti-tumor effect of BCMA CAR-T therapy.
Safety Profile: Manageable Toxicity with Good Tolerability
For elderly, frail, or high-risk MM patients, treatment safety and tolerability are critical for clinical decision-making. In the CAREMM-001 study, BCMA CAR-T therapy exhibited a favorable safety profile with all treatment-related toxicities being manageable, and no treatment-related deaths occurred.
The most common grade 3–4 adverse events were transient cytopenias caused by lymphodepletion chemotherapy, all of which resolved with supportive care, and the median duration of all cytopenias was within 10 days. Immune effector cell-associated hematotoxicity (ICAHT) was predominantly grade 1–2, with only 1 patient developing grade 3 late ICAHT, which improved with blood transfusion and granulocyte colony-stimulating factor support.
CAR-T-specific toxicities were low in incidence and mild in severity: cytokine release syndrome (CRS) occurred in 52.8% of patients, all of which were grade 1–2—far lower than the 75%–80% CRS incidence reported in RRMM patients—and all resolved rapidly with standard management. Immune effector cell-associated neurotoxicity syndrome (ICANS) occurred in only 2 patients (5.6%), both grade 1, which resolved within 1–2 days after glucocorticoid treatment; no patients required potent immunosuppressants such as anakinra.
Infection-related adverse events occurred in 30.6% of patients, with 19.4% being grade ≥3, and pneumonia was the most common infection type (25.0%). Other adverse events were mostly grade 1–2, and no severe adverse events such as tumor lysis syndrome or second primary malignancy were observed. A total of 97.2% of patients completed the planned full course of treatment, confirming the good tolerability of BCMA CAR-T therapy in elderly, frail, and high-risk NDMM patients.
Clinical Significance: A Landmark Study Reshaping the Treatment Landscape
As the world’s first prospective study to evaluate BCMA CAR-T therapy as first-line treatment for transplant-ineligible NDMM patients, the CAREMM-001 study delivers landmark clinical significance and provides critical evidence-based medical data for the expanded clinical application of CAR-T therapy. More importantly, its achievement of 100% MRD negativity and 94.4% CRR far exceeds the historical data of current standard regimens providing the first high-level evidence that the upfront application of BCMA CAR-T therapy in this patient population is not only feasible but also achieves unprecedented deep disease eradication, which is expected to fundamentally alter the existing clinical practice landscape.
Meanwhile, traditional treatment regimens have limited efficacy and yield a poor prognosis for NDMM patients with ultra-high-risk factors such as extramedullary disease, double-hit cytogenetics, and elevated circulating plasma cells. In this study, 37.1% of ultra-high-risk patients achieved deep and durable MRD-negative remission, confirming that BCMA CAR-T therapy can effectively overcome high-risk prognostic factors, break through the efficacy bottleneck of traditional therapies, and provide a powerful new treatment option for this clinically refractory population.
MRD negativity is widely recognized in the academic community as a core prognostic indicator for long-term survival in MM patients. The 100% MRD negativity rate and 15.8-month median follow-up without recurrence achieved in this study bring an unprecedented level of deep remission to transplant-ineligible NDMM patients, making it possible for this population to move toward “functional cure” and shifting the therapeutic concept of multiple myeloma from “long-term disease control” to “pursuing deep molecular remission”.
In addition, the study results support the hypothesis that “earlier application of CAR-T therapy yields superior efficacy”. The use of CAR-T therapy in the early stage of the disease—when tumor burden is low, T cell exhaustion is mild, and immune function is well-preserved—not only achieves a more significant anti-tumor effect but also reduces treatment-related toxicity, providing critical evidence for optimizing the timing of clinical application of CAR-T therapy.
Notably, although ongoing phase III studies such as CARTITUDE-5 and CARTITUDE-6 are evaluating BCMA CAR-T therapy in the NDMM population, there is currently no prospective data for transplant-ineligible patients. The CAREMM-001 study fills this gap by providing key evidence for the first-line application of CAR-T therapy in transplant-ineligible NDMM patients, pioneering the advancement of CAR-T therapy to earlier lines and broader populations (especially high-risk patients) in MM treatment and offering a transformative new treatment option.
Conclusion
The publication of the CAREMM-001 study marks the official entry of BCMA CAR-T therapy into the first-line treatment of transplant-ineligible NDMM patients. This study confirms that frontline BCMA CAR-T therapy enables transplant-ineligible NDMM patients—especially those with high-risk features—to achieve 100% MRD negativity and 94.4% complete response with manageable treatment-related toxicity and good tolerability, successfully overcoming the efficacy bottleneck of traditional therapies and bringing new treatment hope to this clinically refractory population.
These findings not only drive the upgrade of therapeutic concepts for multiple myeloma but also provide new evidence-based medical data for the first-line application of CAR-T therapy in hematologic malignancies. With the conduct of more clinical studies and the optimization of treatment strategies, BCMA CAR-T therapy is expected to further reshape the treatment landscape of multiple myeloma, enabling more patients to achieve deep and durable remission and advancing steadily toward the goal of “functional cure”.
References:
1.Wenqiang, Yan,Chenxing, Du,Rui, Lv et al. Phase II Study of BCMA Chimeric Antigen Receptor T-Cell Therapy in Patients With Newly Diagnosed Multiple Myeloma Ineligible for or Not Proceeding to Autologous Stem-Cell Transplantation (CAREMM-001).[J] .J Clin Oncol, 2026, undefined: JCO2501969.
2. BERDEJA J G, MADDURI D, USMANI S Z, et al. Ciltacabtagene autoleucel, a B-cell maturation antigen-directed chimeric antigen receptor T-cell therapy in patients with relapsed or refractory multiple myeloma (CARTITUDE-1): a phase 1b/2 open-label study [J]. Lancet, 2021, 398(10297): 314-24.
3. Kalariya NM, Hildebrandt MAT, Hansen DK, et al. Clinical outcomes after idecabtagene vicleucel in older patients with multiple myeloma: a multicenter real-world experience. Blood Adv. Sep 10 2024;8(17):4679-4688.
4. Akhtar OS, Oloyede T, Brazauskas R, et al. Outcomes of older adults and frail patients receiving idecabtagene vicleucel: a CIBMTR study. Blood Adv. Apr 8 2025;9(7):1587-1592.
5. MUNSHI N C, ANDERSON L D, JR., SHAH N, et al. Idecabtagene Vicleucel in Relapsed and Refractory Multiple Myeloma [J]. N Engl J Med, 2021, 384(8): 705-16.

Qiu Lugui, M.D., Ph.D.
Professor
Chief Clinical Expert
Institute of Hematology & Blood Diseases Hospital, Chinese Academy of Medical Sciences & Peking Union Medical College
• Chief Physician, Doctoral Supervisor
• Expert Receiving the Special Government Allowance of the State Council
• Distinguished Young and Middle-aged Expert with Outstanding Contributions, National Health Commission
• Member, International Myeloma Society (IMS)
• Committee Member, International Myeloma Working Group (IMWG)
• Editorial Board Member, Blood Advances
• Executive Director, 9th Council of Chinese Anti-Cancer Association (CACA)
• Honorary Chairman, Hematologic Oncology Committee, CACA
• Chairman, Integrated Rehabilitation Committee for Hematologic Oncology, CACA
• Chairman, Myeloma Committee, Chinese Society of Clinical Oncology (CSCO)
• Vice Chairman, 1st Committee of Hematology Institutions Branch, Chinese Hospital Association
• Vice Chairman, 6th Council of Tianjin Anti-Cancer Association
• Editorial Board Member for 6 core journals including Chinese Journal of Hematology
• Completed over 30 major research projects, including National Key Technology R&D Program and National Natural Science Foundation Key Projects
• Published nearly 600 papers, including over 180 SCI-indexed articles
• Edited 6 monographs
• Holds 6 National Invention Patents
• Awarded two First Prizes of Tianjin Science and Technology Progress Award, and three Second Prizes of Science and Technology Progress Award from the Ministry of Education and Tianjin Municipality

An Gang, M.D., Ph.D.
Professor
Institute of Hematology & Blood Diseases Hospital, Chinese Academy of Medical Sciences & Peking Union Medical College
• Chief Physician, Associate Professor, Doctoral Supervisor
• Principal Investigator, State Key Laboratory of Blood and Health
• Young Council Member, Chinese Anti-Cancer Association (CACA)
• Secretary-General, Multiple Myeloma Committee, Chinese Society of Clinical Oncology (CSCO)
• Vice Chairman, 2nd Youth Committee of Hematologic Oncology, Chinese Anti-Cancer Association (CACA)
• Committee Member, Youth Committee, 11th Hematology Society of Chinese Medical Association
• Committee Member, Hematology Translational Medicine Committee, Chinese Anti-Cancer Association (CACA)
• Committee Member, Oncology Nephrology Committee, Chinese Anti-Cancer Association (CACA)
• Secretary, China Multiple Myeloma Research Alliance
• Committee Member, Geriatric Oncology Committee, Tianjin Anti-Cancer Association
• Committee Member, Tianjin Genetic Counseling Committee

Yan Wenqiang, M.D.
• M.D., Resident Physician
• Department of Lymphoma and Myeloma, Institute of Hematology and Blood Diseases Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College
• Research focus: multiple myeloma and rare plasma cell disorders.
• Published 12 papers in peer-reviewed journals including J Clin Oncol, Leukemia, Am J Hematol, J Immunother Cancer, Haematologica, Cancer, among others.
• Delivered oral presentations at ASH and IMS Annual Meetings on multiple occasions. Recipient of IMS Young Investigator Award, ASH Abstract Achievement Award, and Beijing Outstanding Graduate Award.


